Everything You Need To Know About Peptide Therapy For Injury Recovery and Muscle Building
- Harold Pierre, MD

- 2 minutes ago
- 22 min read
A Comprehensive Guide to Using Peptides for Muscle Healing and Tendon Repair
About 9 years ago, I attended an A4M lecture on peptides. I remembered how excited I was to promote these therapies. However, back then, no one knew what I was talking about, and I found very little interests from my patients. Over the last 2-3 years, an explosion of interests has occurred for peptides. Now, I am constantly being asked about peptides. So, I decided to write a very long and comprehensive article about peptides. I threw in everything I can think of in order to cover the common questions. At a later time, I will produce shorter articles for specific peptides.
Why Would You Need Peptides?
Imagine that your rotator cuff has been gnawing at you for six months. Physical therapy helped a little. Cortisone shots bought you a few weeks of relief. And now someone at the gym mentioned peptides for muscle healing and injury recovery, and you're wondering if that's actual science or just another expensive wellness trend dressed up in bro-science.
Fair question. Peptide therapy for tissue regeneration is real medicine, backed by real biology, and increasingly used by physicians who treat sports injuries and musculoskeletal conditions. But the gap between what the science shows and what the internet promises is wide enough to drive a truck through. So let's sort it out.
This guide covers the specific peptides used for tendon repair, muscle repair and cartilage healing. We'll look at how each peptide works at the cellular level, what the actual evidence says and whether taking peptides is worth the cost and risk. Not the hype. The data.
What Are Therapeutic Peptides and Why Do They Matter for Healing?
Peptides are short chains of amino acids that act as the building blocks of larger proteins. Think of them as smaller, more targeted versions of the proteins your body already makes thousands of. These bioactive peptides function as signaling molecules, telling cells what to do, when to grow, when to repair and when to reduce inflammation. The growing interest in peptides in sports medicine and regenerative health comes from this basic biology.
The peptides used in regenerative medicine are either synthetic copies of these natural signals or modified versions designed to last longer in the body. When you tear a tendon or strain a muscle, your body's natural healing response launches a cascade involving growth factors, inflammatory mediators and stem cell recruitment. Peptide therapy aims to supercharge that process and accelerate injury recovery.
That's the theory. And in animal models, it works really well. Certain peptides can stimulate muscle growth, reduce inflammation and speed up the healing process in lab animals. The challenge is that human clinical trials are still catching up.
How to Actually Evaluate a Peptide: The 4-Bucket Framework
Before we get into specific peptides, you need a way to evaluate the claims you'll hear about each one. Peter Attia recently laid out a framework on his podcast (The Drive, AMA #83) that does this well, and it's worth borrowing.
For any peptide, ask five questions. Does it have a plausible biological mechanism? Do we have safety data or dosing protocols from actual studies? Is there evidence it does what people claim in healthy humans? How do the potential risks stack up against the potential benefits? And are there legitimate, FDA-approved alternatives that do the same thing?
Once you run through those questions, most peptides fall into one of Attia's four categories.
Bucket 1 covers peptides with no honest use case. The mechanism is weak or unproven, the data is almost entirely from animal models (often from a single research group) and no human trials exist. Being blunt: if you're being intellectually honest, there's no strong reason to take these.
Bucket 2 includes peptides with a viable mechanism but no clinical trial data. The biology makes sense on paper. But "makes sense on paper" and "works in a human body" are different conversations.
Bucket 3 is where things get more interesting. These peptides have a real mechanism of action plus some safety and efficacy data from human trials. They're not FDA-approved for what people use them for, but at least someone has studied them in actual people.
Bucket 4 is basically gray market copies of FDA-approved drugs. Think compounded versions of semaglutide or tirzepatide sold as "research peptides." These are pharmaceutical knockoffs without the quality controls.
We'll reference this framework as we go through each peptide below. Where a peptide lands in these buckets matters for how you should think about risk.
BPC-157: The Tendon Repair Peptide Everyone's Talking About
Body Protection Compound 157 gets more attention than any other peptide in the musculoskeletal space. It's a 15-amino-acid peptide originally isolated from human gastric juice, and the preclinical data on tendon healing is genuinely impressive.
How BPC-157 Works
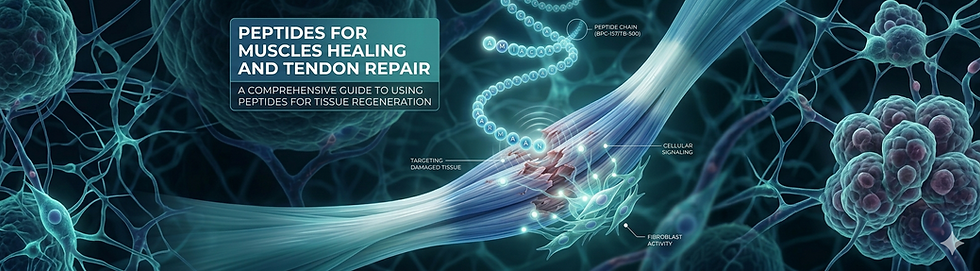
BPC-157 hits multiple repair pathways at once. It ramps up fibroblast activity (the cells that build connective tissue), drives collagen remodeling and boosts angiogenesis through vascular endothelial growth factor (VEGF). New blood supply means more oxygen and nutrients reaching damaged tissue.
It also dials down pro-inflammatory cytokines while activating the PI3K/Akt and TGF-beta signaling pathways. In plain English: it tells your body to build new tissue and stop attacking the injury site with inflammation. It triggers integrin-FAK signaling and nitric oxide release through eNOS, which further supports revascularization and immune regulation at the injury.
What the Research Actually Shows
The animal data is where BPC-157 gets interesting, and where we need to be honest about what we're looking at.
In a 2003 study published in the Journal of Orthopaedic Research, researchers surgically transected Achilles tendons in rats and treated them with BPC-157. The treated tendons showed better biomechanical properties across the board: higher load to failure, improved stiffness and elasticity, better collagen organization and more type I collagen (the strong stuff) compared to controls. The tendons weren't just healing. They were healing stronger.
Rotator cuff injuries have also been studied. A 2014 study presented at the FASEB conference detached the rotator cuffs of 48 rats and gave half of them BPC-157 at 10 mcg/kg. The treated animals showed improved functional recovery and better tendon healing at 2, 4, 8 and 12 weeks compared to saline controls.
Ligament repair tells a similar story. A 2010 study in the Journal of Orthopaedic Research transected the medial collateral ligament (MCL) in rats and tested BPC-157 delivered by injection, topically and orally. All three routes showed consistent functional, biomechanical and histological improvements over 90 days.
There's also evidence that BPC-157 can oppose the tissue-damaging effects of corticosteroids on tendons, which matters because a lot of patients considering peptides have already had multiple cortisone injections.
Now for the caveat that too many peptide blogs leave out: all of this is animal data. Every single study. We hope these results translate to humans, and the biological mechanisms suggest they should. But hoping and proving are different things.
There's another issue that deserves your attention. More than 80% of the published research on BPC-157 comes from a single laboratory. The senior author on most of that work is also listed as an inventor on BPC-related patents and appears as a part-owner of a company whose only drug candidate is BPC-157. That same company sponsored the only registered clinical trial, which has been sitting in "unknown status" for over 11 years with no results posted. Independent replication of these findings is essentially nonexistent.
That doesn't mean the results are fabricated. But in science, replication is everything. When one lab produces all the data and that lab has a direct financial interest in the outcome, you have to factor that in. Peter Attia placed BPC-157 in Bucket 1 of his peptide framework (no honest use case) largely for this reason. That's a harsher assessment than many regenerative medicine physicians would give, but the concern about independent replication is legitimate and you should know about it.
There are also pharmacokinetic questions. Studies in rats and dogs show BPC-157 has a half-life of roughly 30 minutes with a mean bioavailability under 20% in rats and around 40 to 45% in dogs. What that means in humans is still unknown. The full amino acid sequence of the parent BPC protein has never been published, and BPC-157 shows no sequence homology to any known gastrointestinal peptide. That's an unusual origin story for a compound that gets so much clinical attention.
In humans, we have one retrospective case series of 17 patients who received intra-articular BPC-157 knee injections for various types of knee pain over a one-year period. The majority reported symptom improvement. That's encouraging, but 17 patients without a control group isn't the kind of evidence that changes clinical guidelines. It's a starting point, not a conclusion.
Physicians who use BPC-157 in clinical practice often report positive patient outcomes that go beyond what you'd expect from placebo alone. That clinical experience matters. But clinical experience and controlled evidence are two different kinds of knowing, and right now the controlled evidence is thin.
How It's Used
BPC-157 is typically injected subcutaneously or intramuscularly near the injury site. Dosing in the literature ranges from 200 to 1000 mcg per day, and there's no agreed-upon standard. Oral bioavailability exists but appears to be inferior to injection for musculoskeletal applications.
Safety Concerns
A related compound called pentadecapeptide arginate carries a theoretical risk of encouraging tumor growth in dysplastic tissues. Some evidence actually suggests the opposite, that it may inhibit tumor growth. But this is exactly the kind of uncertainty that makes physician guided peptide protocols so important. You don't want to guess your way through this.
TB-500: How This Peptide Works for Muscle Repair
Thymosin Beta-4 is a protein your body naturally produces in higher amounts after tissue injury. TB-500 is a synthetic fragment of that protein, and it's become a staple in medical peptide therapy for sports injuries. Peptides like TB-500 are popular among athletes looking for performance and recovery support, though the evidence base is still limited to animal data.
Mechanism of Action
TB-500 drives actin polymerization, which is the basic building block of cellular structure and movement. It recruits progenitor cells (basically repair cells) to the injury site and enhances cellular migration so those cells can actually get where they need to go.
The anti-inflammatory effects mirror what we see with BPC-157, and the proangiogenic activity is comparable. TB-500 acts on the MAPK, NF-kB and FAK pathways, supporting tissue healing and reducing the destructive phase of inflammation.
Clinical Evidence
A 1999 study in the Journal of Cell Biology showed that thymosin beta-4 increased wound reepithelialization by 42 to 61% in rats, with a two to threefold increase in keratinocyte migration and increased collagen deposition. Later research showed that muscle injury specifically upregulates thymosin beta-4 expression, suggesting the body already uses this molecule as a repair signal. The peptide increases cell migration, angiogenesis and stem cell mobilization in animal models.
Racehorses have been treated with thymosin beta-4 for tendon injuries for years with positive results, and that veterinary track record is what prompted interest in human musculoskeletal applications.
But again, we're looking at animal models and veterinary data. No controlled human trials exist for TB-500 in muscle or tendon repair. The mechanisms are well-understood and the preclinical results are consistent. We're waiting for human studies to confirm what the animal data strongly suggests.
Administration
TB-500 is given subcutaneously or intramuscularly. Oral bioavailability is limited, so injection remains the primary route for muscle recovery applications.
GHK-Cu: The Collagen and Wound Healing Peptide for Tissue Remodeling
Glycyl-Histidyl-Lysine-Copper is a naturally occurring tripeptide that binds copper. It's been used in dermatology and aesthetic medicine for years, and now it's drawing attention for tissue regeneration in the musculoskeletal world.
How GHK-Cu Supports Repair
GHK-Cu stimulates dermal fibroblast proliferation and regulates matrix metalloproteinases (MMPs), the enzymes responsible for breaking down and rebuilding the extracellular matrix. This is critical for tissue remodeling. You don't just want scar tissue filling a gap. You want organized, functional tissue.
It also provides antioxidant effects through the NRF2 pathway and the copper-redox axis, reducing oxidative damage at the injury site. For soft tissue injuries where scar modulation matters, GHK-Cu offers something the other peptides don't.
Current Applications
The strongest evidence comes from skin healing and cosmetic applications. In the orthopaedic space, one 2015 study in the Journal of Orthopaedic Research tested GHK-Cu in a rat ACL reconstruction model. At six weeks, treated rats showed significantly less knee laxity and higher stiffness compared to controls. But here's the catch: by 12 weeks, those benefits had faded. No significant difference in laxity, load to failure or gait between groups.
That transient benefit is an important finding. It tells us GHK-Cu may help during the early healing window but might not sustain its effects long-term without continued treatment. More research is needed, and like the other peptides here, we're still in the animal model phase for musculoskeletal applications. The biological rationale for Glycyl Histidyl Lysine Copper for tissue remodeling is sound, but human trials haven't happened yet. This peptide is typically administered subcutaneously.
Growth Hormone-Releasing Peptides: How They Build Muscle and Cartilage
This category includes several peptides that stimulate your pituitary gland to produce more human growth hormone naturally. Unlike injecting synthetic growth hormone directly (which shuts down your body's own production), growth hormone secretagogues like ipamorelin and CJC-1295 preserve the normal feedback loop. The use of peptides in this class is one of the more evidence-backed approaches in sports medicine.
They work through the GH/IGF-1 axis, activating the PI3K/Akt/mTOR cascade. That's the master switch for muscle protein synthesis, satellite cell activation and cartilage matrix production. For anyone trying to build muscle or improve muscle strength after an injury, this pathway matters.
Ipamorelin
Ipamorelin is a selective ghrelin receptor agonist. It triggers growth hormone release by inhibiting somatostatin (the hormone that tells your pituitary to stop making GH). What makes ipamorelin stand out is its selectivity. It doesn't spike cortisol or prolactin the way other GH-releasing peptides can. Fewer side effects, cleaner hormonal profile.
CJC-1295
CJC-1295 is a long-acting growth hormone-releasing hormone (GHRH) analog. Published human data shows dose-dependent increases in growth hormone (2 to 10 fold) and IGF-1 (1.5 to 3 fold) with sustained effects. It comes with or without a drug affinity compound (DAC) that affects how long the GH elevation lasts. The version without DAC (often called Modified GRF 1-29) paired with ipamorelin has become one of the most popular physician prescribed peptide therapy combinations. Together, they create a pulsatile rise in GH and IGF-1 that mimics your body's natural rhythm. The individual compounds each have published data, though the specific combination hasn't been tested in human trials.
Tesamorelin
Tesamorelin is a 44-amino-acid GHRH analog that's actually FDA-approved for HIV-related lipodystrophy. Its established safety profile has led physicians to explore off-label use for musculoskeletal recovery. The anabolic effects on lean body mass are well-documented, and this is one of the few peptides in this space with real regulatory backing.
Sermorelin
Another GHRH analog, sermorelin is FDA-approved for children with growth hormone deficiency. Like tesamorelin, it's used off-label in adult musculoskeletal recovery. These are among the few peptides where we have decades of safety data.
What GH Secretagogues Do for Injury Recovery
The recovery benefits for accelerated healing after orthopaedic surgery are where this class gets interesting. Growth hormone secretagogues may activate satellite cells (the stem cells of muscle tissue) and boost myofibrillar protein synthesis. They can help mitigate disuse atrophy, which is the muscle wasting that happens when you're immobilized after surgery or injury. For patients trying to maintain lean muscle mass during a recovery process, peptide therapy might offer real advantages here.
Elevated insulin-like growth factor-1 (IGF-1) is linked to better chondrocyte proliferation, bone healing and fracture repair. There's also a sleep quality benefit. GH is primarily released during deep sleep, and these peptides tend to improve sleep architecture. Better sleep means faster recovery. Period.
AOD-9604: The Injectable Peptide for Cartilage Repair and Joint Pain
AOD-9604 is a synthetic fragment of human growth hormone that was originally developed as an anti-obesity drug. Its orthopaedic relevance comes from its effects on cartilage repair without the metabolic complications of full-length growth hormone.
Why AOD-9604 Is Different
Unlike other GH-related peptides, AOD-9604 doesn't significantly alter glucose levels or systemic IGF-1. A meta-analysis of six randomized controlled trials confirmed this safety profile. That matters for patients with metabolic concerns who still need tissue repair support.
A 2015 study in Annals of Clinical and Laboratory Science tested AOD-9604 in a rabbit knee osteoarthritis model. Researchers gave 32 rabbits with collagenase-induced OA weekly injections of either saline, hyaluronic acid alone, AOD-9604 alone or the combination. The AOD-9604 plus hyaluronic acid group showed significantly better cartilage morphology and histological scores than either treatment alone, with shorter lameness periods.
One study in rabbits isn't a clinical guideline. But it's the only published data we have on this combination, and the results were positive enough that physicians exploring medical management of tissue repair are increasingly pairing AOD-9604 with hyaluronic acid or platelet-rich plasma (PRP) in regenerative protocols for osteoarthritis. Human trials are needed to confirm whether these rabbit results hold up in our joints.
Sleep and Injury Recovery Peptides: How Peptide Therapy Works While You Rest
Healing doesn't happen in the gym or even in the doctor's office. Most tissue repair occurs during deep sleep when growth hormone secretion peaks and inflammatory processes settle down. Several peptides target this recovery window directly.
Epithalon
Epithalon is a synthetic tetrapeptide derived from the pineal gland peptide epithalamin. It regulates melatonin production, normalizes circadian rhythms and activates telomerase (the enzyme that maintains chromosome stability). It also influences SIRT1 and CLOCK gene pathways, enhances endogenous GH secretion and reduces oxidative stress.
The longevity medicine crowd loves epithalon, but the orthopaedic benefit is indirect and real: better sleep and better hormonal regulation mean faster, more complete tissue repair.
DSIP (Delta Sleep-Inducing Peptide)
DSIP is a neuropeptide found in the hypothalamus that triggers delta wave sleep in rodent models. It may increase endogenous GH secretion and modulate stress-related hormonal responses. For patients recovering from surgery or injury, optimizing sleep quality can meaningfully accelerate the timeline.
Pinealon
This tripeptide supports mitochondrial function and neuronal metabolism. It enhances cognitive function, improves sleep quality and builds resilience to oxidative stress, particularly in aging or stressed neural tissues. For older patients going through rehabilitation, pinealon may support both the cognitive demands and the physical recovery process.
Neurological Recovery Peptides: Mind-Muscle Connection
Rehabilitation after a major injury or surgery isn't purely physical. Fear of re-injury, anxiety about returning to sport and the mental fatigue of prolonged recovery all affect outcomes. Several peptides address the neurological side of healing.
Selank
Selank is a synthetic heptapeptide based on the immunomodulatory molecule tuftsin. It's approved in Russia for anxiety treatment. It modulates serotonin and dopamine, increases brain-derived neurotrophic factor (BDNF) and enhances synaptic plasticity. For patients struggling with the psychological burden of prolonged recovery, selank can support mental clarity and emotional regulation without the side effect profile of traditional anxiolytics.
Semax
Semax is a synthetic neuropeptide derived from adrenocorticotropic hormone (ACTH), used in Russia for neurological conditions including ischemic stroke. It upregulates BDNF, stimulates synaptogenesis and provides neuroprotective effects. For prolonged neuromuscular retraining, peripheral nerve recovery or simply maintaining focus during months of rehabilitation, semax offers therapeutic peptides for musculoskeletal healing support that most people don't think about.
Dihexa
Originally developed for cognitive decline in neurodegenerative diseases, dihexa potentiates hepatocyte growth factor (HGF) activity and stimulates the c-Met receptor pathway. It stimulates synaptogenesis, supports neural remodeling and may improve the mind-muscle connection during rehabilitation. This one is early-stage, but the mechanism is interesting for patients doing complex motor retraining.
How These Peptides Work Together: Using Peptides for the Healing Process
Understanding how peptides work at the cellular level explains why peptide therapy for tissue regeneration generates so much clinical interest. These molecules target overlapping but distinct biological pathways. Here's a simplified map:
For tissue building and repair, PI3K/Akt drives fibroblast proliferation and collagen synthesis, mTOR activates muscle protein synthesis and TGF-beta remodels the extracellular matrix.
On the inflammation side, NF-kB resolution calms the inflammatory cascade while MAPK supports tissue healing and manages the immune response.
For blood supply, VEGF drives angiogenesis, integrin-FAK signaling supports revascularization and eNOS releases nitric oxide for vasodilation.
Energy and metabolism matter more than people realize because damaged tissue is metabolically hungry. AMPK improves mitochondrial efficiency to meet that demand.
And for neural recovery, HGF/c-Met supports neuroplasticity and neuromuscular recovery while BDNF via TrkB receptors supports synaptic growth.
When a physician designs a peptide protocol, they're often stacking peptides that hit different nodes in this network. BPC-157 and TB-500 for direct tissue repair. A GH secretagogue for anabolic support. Maybe GHK-Cu for tissue remodeling. And a sleep peptide to improve the recovery window. That stacked approach is what makes cellular medicine peptide protocols different from taking a single pill.
Why Most Oral Peptides Don't Work the Way You Think
You'll see oral peptide products everywhere online. Capsules, sublingual drops, sprays. The marketing makes it sound simple. But the biology tells a different story.
Peptides are chains of amino acids. Your digestive system is designed to break amino acid chains apart. That's literally what digestion does. So when you swallow a peptide, your gut enzymes chew it up before it ever reaches your bloodstream in its intact, functional form.
The pharmaceutical industry has spent billions trying to solve this problem. Even with the most advanced delivery engineering money can buy, the results are humbling. Oral semaglutide (the FDA-approved GLP-1 drug) achieves a bioavailability somewhere between 0.4 and 1% compared to roughly 89% for the injectable version. That's after years of pharmaceutical optimization with specialized absorption enhancers and precise formulation work.
If a massive pharma company can only get 1% oral bioavailability for a drug they've spent billions developing, what are the chances that a gray market oral peptide capsule is doing better? The answer isn't encouraging.
There are exceptions. Collagen peptides work orally because they function as a substrate and signaling stimulus after digestion. They don't need to arrive intact at a specific receptor. But most therapeutic peptides require intact receptor interaction to do their job, and they simply don't survive digestion in meaningful amounts.
If your peptide protocol involves injection, that's the route with actual pharmacological support behind it. If someone is selling you oral BPC-157 or oral TB-500 at a premium, ask them for the bioavailability data. You'll get a long pause.

Safety: What You Need to Know Before Starting Peptide Therapy
Injectable peptides are generally considered safe in the short term based on available data. The adverse effect profile of therapeutic peptides is favorable compared to many conventional treatments. Most commonly reported side effects are minor and self-limiting: injection site redness, transient hormonal shifts and occasional receptor desensitization with prolonged use.
But "generally favorable" is not the same as "proven safe." And that distinction matters.
The Animal-to-Human Gap
A 2025 systematic review of BPC-157 in orthopaedic sports medicine found 36 studies on the topic. Thirty-five were preclinical (animal models). One was a small human case series. That ratio tells you everything you need to know about where we are with the evidence.
The animal studies are consistent and the mechanisms make biological sense. Rat tendons heal faster and stronger. Rabbit cartilage looks better on histology. Mouse wounds close quicker. These aren't trivial findings. But the history of medicine is full of treatments that worked beautifully in animals and failed in humans. We should be optimistic about what these peptides can do, but honest that we're still early in proving it.
Current orthopaedic literature on peptides is dominated by animal models, small prospective cohorts and case series. Randomized controlled trials are scarce. The evidence gap is especially wide for musculoskeletal-specific outcomes like tendon-to-bone healing and cartilage repair.
Dosing varies wildly between studies. Routes, frequencies and durations are all over the map. BPC-157 studies use anywhere from 200 to 1000 mcg per day with no consensus on what's optimal. This isn't a reason to avoid peptides entirely, but it is a reason to work with a physician who understands the literature and can make informed dosing decisions.
Quality Control and the Gray Market Problem
Not all peptides are created equal. Variability in purity, potency and sterility is a documented problem with compounded peptides. Inconsistent dosing, label discrepancies and endotoxin contamination (lipopolysaccharides) have all been reported.
Most of the peptides discussed in this article are sold through what's called the gray market. They're labeled "for research use only" and shipped directly to consumers or clinics without the manufacturing oversight that FDA-approved drugs require. That label isn't a technicality. It means nobody is verifying that what's in the vial matches what's on the label with any consistency.
Some people point to independent third-party testing as a safeguard. And testing can reduce uncertainty about identity and purity for the specific vial that was tested. But it can't certify sterility or endotoxin levels across all vials, lots and storage conditions. Sampling is sporadic. Peptides are chemically fragile. A test result from one batch tells you very little about the next batch.
Peptides are also expensive to manufacture properly. The same economic pressures that make FDA-approved peptide drugs like semaglutide and tesamorelin cost what they do apply to gray market production. When a gray market product costs a fraction of the pharmaceutical version, something was cut. Sometimes it's quality control. Sometimes it's purity. Sometimes it's sterility testing. You won't always know which.
If you're considering peptide therapy, your provider should be sourcing from cGMP-compliant suppliers with FDA inspection history. Ask about certificates of analysis, third-party sterility testing, endotoxin testing and potency verification. If a provider can't tell you where their peptides come from and how they're tested, find a different provider.
Regulatory Status
Most peptides discussed in this article are not FDA-approved for orthopaedic use. Tesamorelin and sermorelin have FDA approval for other conditions, which provides safety data that supports off-label musculoskeletal applications. The rest are available through compounding pharmacies under sections 503A and 503B of the Federal Food, Drug, and Cosmetic Act.
Recent FDA actions have placed some peptides on interim category lists that restrict their use in compounding unless further studied. The regulatory picture is evolving, and a physician who stays current on these changes is essential.
A Note for Athletes
Many of these peptides are banned by the World Anti-Doping Agency. Tesamorelin, ipamorelin, CJC-1295, sermorelin and others are prohibited for competitive athletes. If you're subject to drug testing, this matters. For the general population seeking tissue repair under medical supervision, these restrictions don't apply, but you need to know they exist.
Who Is a Good Candidate for Peptide Therapy?
Peptide therapy isn't for everyone, and it shouldn't be the first thing you try. Physical therapy, proper nutrition, adequate sleep and time are still the foundation of musculoskeletal recovery. But for patients who have plateaued with conventional treatments, or who are looking for accelerated healing after orthopaedic surgery, peptides may offer a layer of biological support that other treatments don't provide.
Good candidates typically include people dealing with chronic pain from tendon injuries that haven't responded to conservative treatment, athletes in sports medicine programs looking for faster recovery timelines, patients with chronic joint pain or early-stage osteoarthritis exploring regenerative medicine options and individuals experiencing age-related decline in tissue repair capacity. If you're dealing with persistent joint pain, swelling and pain that won't quit or muscle mass loss from prolonged immobilization, peptide therapy might be worth discussing with your doctor.
This should always be physician prescribed peptide therapy. A telemedicine peptide consultation can be a good starting point, but the provider needs to understand your full medical history, current medications and specific injury before designing a protocol. Hormone replacement and peptide treatments often overlap, and those interactions need to be managed by someone who knows what they're doing.
What to Expect from a Peptide Protocol
Realistic expectations matter here. Peptides aren't magic. They don't replace the need for rehabilitation, and they won't fix a problem that requires surgery. Peptides such as BPC-157 and TB-500 are tools, not miracles.
What they can do is create a more favorable biological environment for healing. Patients on well-designed protocols typically report reduced pain and less inflammation within the first few weeks, improved recovery timelines (though individual responses vary significantly), better sleep quality with GH secretagogue protocols, gradual improvements in muscle strength and tissue function over 8 to 16 weeks, pain relief during rehabilitation and better tolerance for physical therapy exercises.
Peptides help create the conditions for repair, but you still have to do the work. The timeline depends on the injury, the specific peptides used and individual factors like age, overall health and compliance with the full treatment plan. Peptide therapy works best as part of a full approach that includes physical therapy, nutrition and lifestyle changes.
Finding a Qualified Provider
If you're interested in peptide therapy, finding the right provider is the most important decision you'll make. Peptides currently sit in a gray area between experimental and mainstream, and the field is moving fast. Look for physicians who specialize in regenerative medicine, understand the literature on therapeutic peptides for musculoskeletal healing and can explain both the potential benefits and the limitations of current evidence.
Regenerative medicine practices in Dallas Texas, Scottsdale Arizona and other major metro areas increasingly offer peptide protocols as part of their treatment menu. Telemedicine has expanded access significantly. Wherever you are, the key is finding a provider who treats peptides as serious medicine, not a wellness trend.
Ask them about their sourcing, their dosing rationale and how they monitor patient outcomes. A good provider will be honest about what we know and what we don't. That honesty is more valuable than a sales pitch.
Frequently Asked Questions
What are the most studied peptides for tendon repair and muscle recovery?
BPC-157 and TB-500 are the two most studied peptides for direct tissue repair. BPC-157 has the strongest preclinical evidence for tendon healing, with rodent studies showing improved biomechanical strength and a small human case series showing symptom reduction. TB-500 has extensive veterinary data supporting its use in muscle and tendon repair. Growth hormone secretagogues like ipamorelin and CJC-1295 support recovery indirectly through the GH/IGF-1 axis.
Are peptides FDA-approved for treating sports injuries or musculoskeletal conditions?
Most therapeutic peptides used for tissue repair are not FDA-approved for orthopaedic indications. Tesamorelin is FDA-approved for HIV-related lipodystrophy and sermorelin for pediatric growth hormone deficiency, but their musculoskeletal use is off-label. Other peptides are available through compounding pharmacies. Working with a knowledgeable physician who understands the regulatory landscape and evidence base is essential for safe, informed use.
Can peptide therapy replace surgery for tendon or ligament injuries?
No. Peptide therapy doesn't replace surgical intervention when surgery is indicated. A complete tendon rupture or unstable ligament tear still requires surgical repair. Peptides may support healing after surgery by creating a more favorable biological environment for tissue regeneration, potentially improving recovery timelines and outcomes. They're best used as part of a full medical management of tissue repair plan that includes physical therapy and appropriate conventional treatments.
How do I find a qualified physician for peptide therapy?
Look for providers specializing in regenerative medicine or sports medicine who have experience with physician guided peptide protocols. Ask about their peptide sourcing (it should be from cGMP-compliant facilities), how they determine dosing, and how they monitor outcomes. Many providers now offer telemedicine peptide consultation for initial evaluations. Avoid any provider who can't discuss the evidence honestly or who presents peptides as guaranteed solutions.
Do oral peptides work for muscle and tendon repair?
Most oral peptides have very poor bioavailability because your digestive system breaks them down before they reach the bloodstream intact. Even highly engineered pharmaceutical peptides like oral semaglutide achieve less than 1% bioavailability compared to injectable forms. Collagen peptides are an exception because they work as a substrate after digestion rather than needing to bind a specific receptor. For therapeutic peptides like BPC-157 and TB-500, injection remains the only route with meaningful pharmacological support.
How do you evaluate whether a peptide is worth trying?
A useful approach is to ask five questions about any peptide: Does it have a proven biological mechanism? Is there human safety data? Does evidence show it works in healthy humans (not just animals)? Do benefits outweigh the risks? And are there FDA-approved alternatives? Most gray market peptides fall short on at least two or three of these criteria. The peptides with the strongest cases for musculoskeletal use are those with some human trial data and FDA-approved relatives in the same drug class, like the growth hormone secretagogues.
References
Therapeutic Peptides in Orthopaedics: Applications, Challenges, and Future Directions. PMC/National Library of Medicine. PMC12753158. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC12753158/
Staresinic M, et al. Gastric pentadecapeptide BPC 157 accelerates healing of transected rat Achilles tendon and in vitro stimulates tendocytes growth. Journal of Orthopaedic Research. 2003. PMID: 14554208.
Sikiric P, et al. Effect of pentadecapeptide BPC 157 on rotator cuff tear injury in rat. The FASEB Journal. 2014;28(S1):844.9.
Cerovecki T, et al. Pentadecapeptide BPC 157 (PL 14736) improves ligament healing in the rat. Journal of Orthopaedic Research. 2010. PMID: 20225319.
Malinda KM, et al. Thymosin beta4 accelerates wound healing. Journal of Investigative Dermatology. 1999. PMID: 10469335.
Fu SC, et al. Tripeptide-copper complex GHK-Cu (II) transiently improved healing outcome in a rat model of ACL reconstruction. Journal of Orthopaedic Research. 2015. PMID: 25731775.
Kwon DR, et al. Effect of Intra-articular Injection of AOD9604 with or without Hyaluronic Acid in Rabbit Osteoarthritis Model. Annals of Clinical and Laboratory Science. 2015. PMID: 26275694.
Lee E, Padgett B. Intra-Articular Injection of BPC 157 for Multiple Types of Knee Pain. Alternative Therapies in Health and Medicine. 2021. PMID: 34324435.
Emerging Use of BPC-157 in Orthopaedic Sports Medicine: A Systematic Review. PMC/National Library of Medicine. 2025. PMC12313605.
Regeneration or Risk? A Narrative Review of BPC-157 for Musculoskeletal Healing. PMC/National Library of Medicine. 2025. PMC12446177.
Local and Systemic Peptide Therapies for Soft Tissue Regeneration: A Narrative Review. PMC/National Library of Medicine. 2024. PMC11426299.
Teichman SL, et al. Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I secretion by CJC-1295, a long-acting analog of GH-releasing hormone, in healthy adults. Journal of Clinical Endocrinology and Metabolism. PMID: 16352683.
Pickart L, et al. Regenerative and Protective Actions of the GHK-Cu Peptide in the Light of the New Gene Data. International Journal of Molecular Sciences. 2018. PMC6073405.
Attia P. AMA #83: Peptides: evaluating the science, safety, and hype in a rapidly growing field. The Peter Attia Drive. Episode #387. April 13, 2026. Available at: https://peterattiamd.com/ama83/
Pharmacokinetics, distribution, metabolism, and excretion of body-protective compound 157, a potential drug for treating various wounds, in rats and dogs. Frontiers in Pharmacology. 2022. PMID: 36588717.
About the Author
Harold Pierre, MD, is a board-certified anesthesiologist, board-certified addiction medicine specialist, and a concierge addiction doctor based out of Tulsa, Oklahoma with over 27 years of experience. He is board-certified by the American Board of Anesthesiology and the American Board of Preventive Medicine, and has extension experience managing hormones, pain, addiction, and their intersection. He is licensed in Florida, Texas, Oklahoma, South Carolina, Louisiana, and Arizona. If you are seeking care, you may schedule an appointment with him by calling or texting 918-518-1636. LinkedIn
Disclaimer
This article is for informational purposes only and does not constitute medical advice. Peptide therapy should only be pursued under the supervision of a qualified physician who can evaluate your individual health needs and circumstances. Many of the peptides discussed are not FDA-approved for musculoskeletal conditions, and the evidence base is still developing. Always consult with your healthcare provider before starting any new treatment. To schedule a consultation, visit frugaldoctor.com/consultation.





Comments